Our customers work more efficiently and benefit from
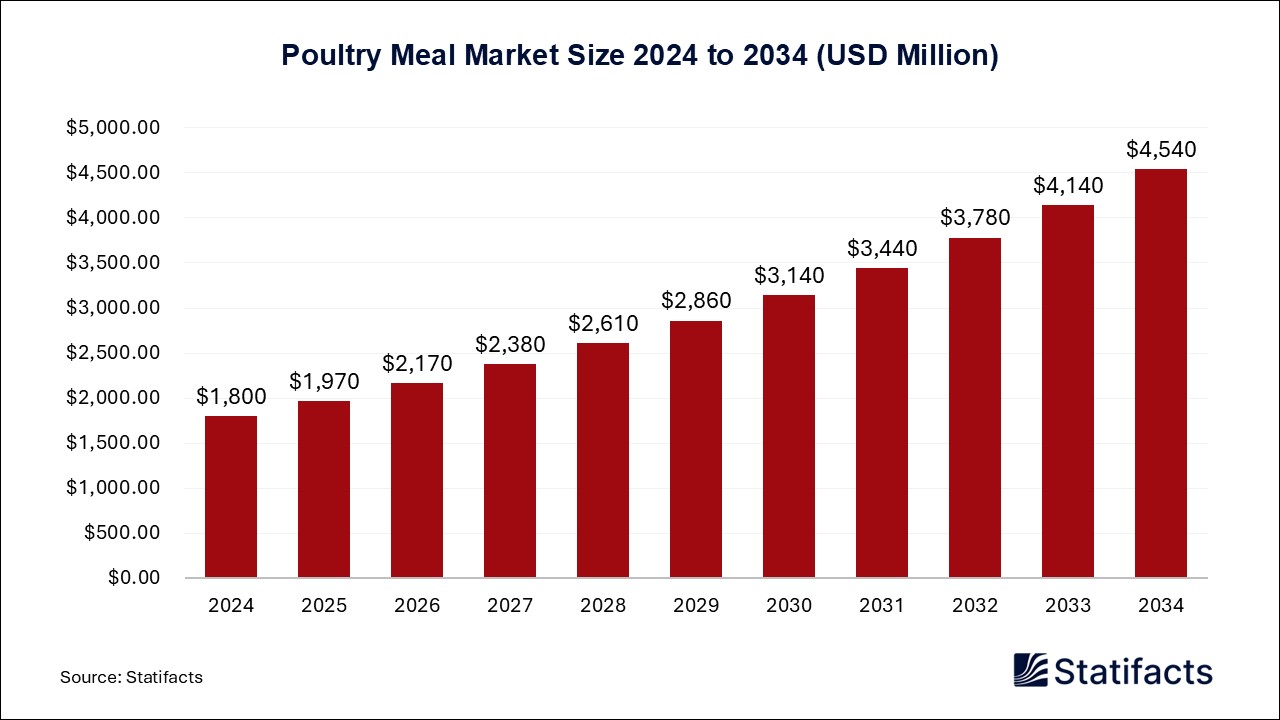
The global eClinical solutions market size surpassed USD 10,730 million in 2024 and is predicted to reach around USD 40,500 million by 2034, registering a CAGR of 14.2% from 2025 to 2034.
| Industry Worth | Details |
| Market Size in 2025 | USD 12,260 Million |
| Market Size by 2034 | USD 40,500 Million |
| Market Growth Rate from 2025 to 2034 | CAGR of 14.2% |
The eClinical solutions market refers to the production, distribution, and application of eClinical solutions, which are a range of technology-driven tools, platforms, and services that are used in the management and conduct of clinical trials. These solutions aim to streamline and improve many aspects of the clinical trial process, including data collection, management, analysis, monitoring, and reporting. eClinical solutions bring people & data together to support tomorrow's breakthroughs with clinical data software and clinical biometrics services. eClinical Solutions’ data & analytics platform and biometrics services experts help biopharmaceutical researchers around the world manage trial complexity faster.
eClinical’s technology-driven approach provides medical monitors with one central source of truth across data sources, allowing collaboration, real-time oversight, enhanced quality, and a richer understanding of the patient experience. eClinical solution benefits include reduced cycle times, enhanced productivity, easy scale, and develop tomorrow's breakthroughs with today’s resources. Digital solutions refer to technology-based tools and services designed to improve processes, enhance efficiency, and solve specific problems in many sectors. These solutions leverage digital technology to create streamlined workflows, facilitate communication, and foster innovation.
Rising R&D expenditure across the globe is driving the growth of the eClinical solutions market. Increasing investment in R&D benefits include increased market participation, cost management benefits, innovations in marketing abilities, and trend-matching are all reasons companies invest in R&D. R&D expenditure drives innovation and technological progress, directly contributing to GDP improvement by creating innovative products, processes, and knowledge spillover across industries. R&D can lead to discoveries and ideas, or the invention or enhancement of products and services. R&D is the first step to creating something new.
Research and development (R&D) is an essential driver of economic growth as it spurs innovation, invention, and progress. R&D spending can lead to breakthroughs that can drive profits and well-being for consumers. A research and development (R&D) engineer creates new and refines existing, products and technologies through a process of researching, designing, experimenting, and testing. Their duties include creating and developing new products to meet consumer needs.
Rising adoption of software solutions in clinical trials is driving the growth of the eClinical solutions market. Software solutions minimize human errors and improve data integrity. The initial investment can be significant, but the long-term savings due to enhanced efficiency, reduced need for manual labor, and fewer errors make clinical trial software a cost-effective solution. A clinical trial management system is an integrated cloud-based software platform that is used for the end-to-end management of clinical trials.
A clinical trial management system is used to plan, track, and analyze clinical trials. It also finds and manages participating patients. The purpose of research software as a component in our instruments can include acquisition, methods to stream or upload experimental data, data cleaning, and processing. Generally, research software components organize, serve, and provide access to data. Software is a set of instructions, data, or programs used to operate computers and execute specific tasks. It helps to perform many functions, such as running operating systems, browsers, and productivity software like Microsoft Office and email clients.
Increasing demand for outsourcing clinical trials to CROs is driving the growth of the eClinical solutions market. By working with a CRO, drug companies can outsource some or all of the clinical trial management activities, reducing the need for in-house resources and expertise. In addition to reducing strain on internal resources, CROs have the infrastructure and resources necessary to conduct clinical trials efficiently. CRO outsourcing is the process of hiring an outside company or individual to manage the company’s conversion rate optimization efforts.
This can include managing CRO strategy, conducting consumer research, running A/B tests, and more. Clinical outsourcing is an alternative to keeping the process in-house. Clinical outsourcing has long been a popular cost-saving measure. The potential benefits of outsourcing are significant. It allows you to tap into global talent, reduce costs, and enhance operational efficiency, which can be a game changer for business.
Published by Laxmi Narayan , March 2025
For any questions about this dataset or to discuss customization options, please write to us at sales@statifacts.com
| Stats ID: | 8121 |
| Format: | Databook |
| Published: | March 2025 |
| Delivery: | Immediate |
| Price | US$ 1550 |


| Stats ID: | 8121 |
| Format: | Databook |
| Published: | March 2025 |
| Delivery: | Immediate |
| Price | US$ 1550 |

You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.
Unlock unlimited access to all exclusive market research reports, empowering your business.
Get industry insights at the most affordable plan
Stay ahead of the competition with comprehensive, actionable intelligence at your fingertips!
Learn More Download
Download